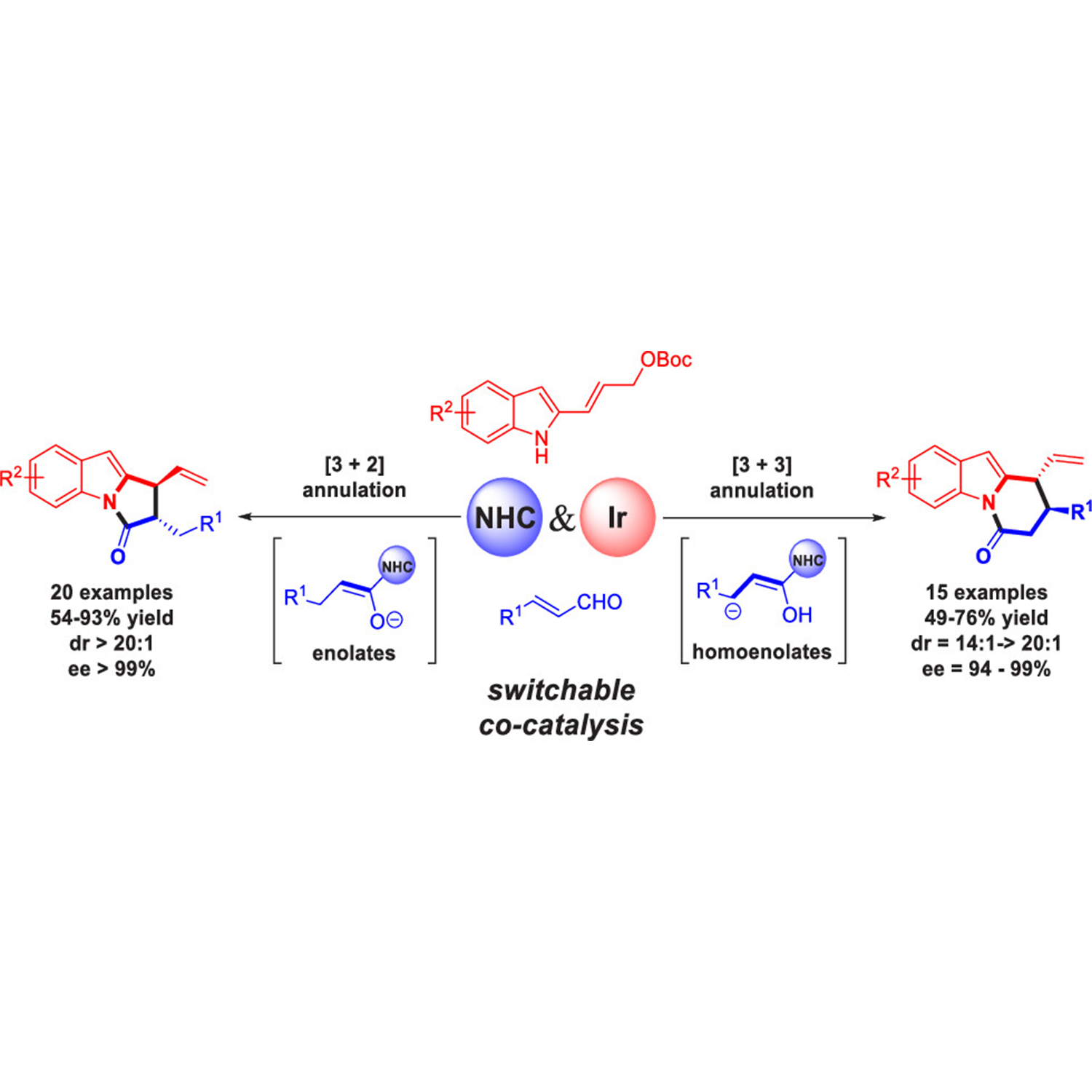
ABSTRACT: A cooperative N-heterocyclic carbene (NHC)/iridium catalysis has been developed to achieve highly stereoselective and regiodivergent [3 + 2] and [3 + 3] annulation reactions of 2-indolyl allyl carbonates with enals. The use of the NHC catalyst has introduced switchable homoenolate and enolate intermediates from the common enal precursor via a simple adjustment of reaction conditions in a predictable manner. This protocol furnishes two types of biologically important products, pyrrolo[1,2-a]indoles and pyridine[1,2-a]indoles, with high diastereo- and enantioselectivities (up to >20:1 dr and >99% ee). Notably, all four stereoisomers of these products with two vicinal stereocenters could be afforded through permutations of the enantiomers of the two chiral catalysts. Mechanistic investigations and further computational density functional theory calculations give an explanation of the origin of the regioselectivity. In addition, the NHC-enolate intermediate generated from formylcyclopropanes was also compatible in this cooperative catalytic system and thus the arsenal of optically pure pyrrolo[1,2-a]indole products was enriched.
For detail:https://pubs.acs.org/doi/abs/10.1021/acscatal.1c00081